Dr. Pen M9 Backflow Prevention Verification – BuyDermaRollers.com
In the medical aesthetics and skincare industry, the “backflow prevention” feature of microneedling devices is often mentioned—but is it truly a safety design, or just a marketing claim?
In real-world use, many people are concerned about whether serums can flow back into the device, and whether it is genuinely safe. To address these questions, we conducted a straightforward test to demonstrate the actual performance of the Dr. Pen M9.
Dr. Pen M9 is an electric microneedling device designed for intuitive operation and professional-level precision. This intelligent system represents a new evolution in microneedling technology. Designed with intelligent control at its core, this advanced device simplifies treatment adjustment while delivering consistent, precise, and reliable performance across different skin areas. It is designed for those seeking professional-grade results at home, the Dr. Pen M9 delivers intelligent, consistent performance with every use.

1. Why is “backflow prevention” so important?
During microneedling procedures, if serum flows back through the needle into the device, it may lead to two major risks:
– On one hand, residual liquid can easily cause cross-contamination. Even if a new cartridge is used, the risk of secondary infection cannot be completely eliminated.
– On the other hand, if liquid enters the motor, it may cause corrosion or even short circuits, directly affecting the device’s stability and potentially leading to equipment damage.

2. Dr. Pen M9 Backflow Prevention Structure Analysis
The Dr. Pen M9 has been structurally upgraded to specifically reduce the risk of liquid backflow at the source.
Its core improvement lies in the newly optimized cartridge design, particularly the integrated silicone stopper at the rear. Made from a highly elastic sealing material, it tightly wraps around the drive shaft, maintaining excellent sealing performance even during high-speed operation—effectively preventing serum or bodily fluids from flowing back into the device.
This design not only enhances safety during use, but also improves the device’s stability and durability, allowing it to maintain consistent performance even in high-frequency usage scenarios.
3. Test Section: Backflow Prevention Verification
To verify the Dr. Pen M9’s actual protective performance, we simulated a high-intensity, real-world operating environment.
– Test Method: Diluted red ink was added into a container to simulate a serum environment. After installing the M9, the device was operated at the highest speed setting, allowing the cartridge to run continuously. During the process, the angle of the device was constantly adjusted to replicate real-world skincare application scenarios.

– Test Observation: The device operated continuously and stably in the liquid environment, with the cartridge maintaining high-speed performance throughout. No abnormal interruptions or signs of liquid backflow were observed.

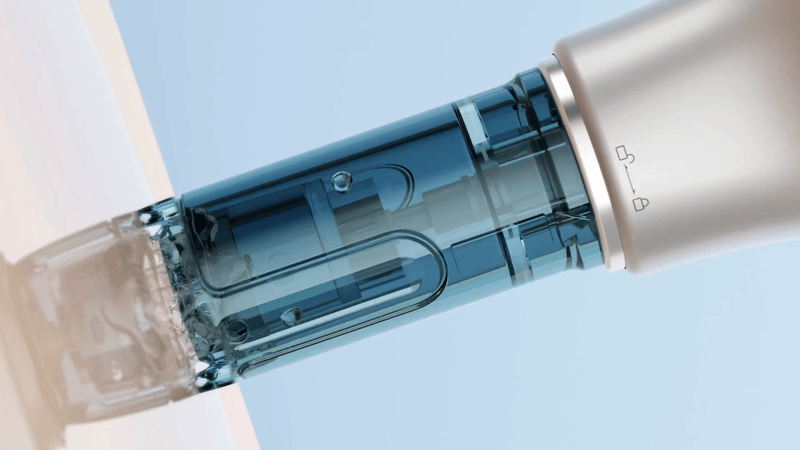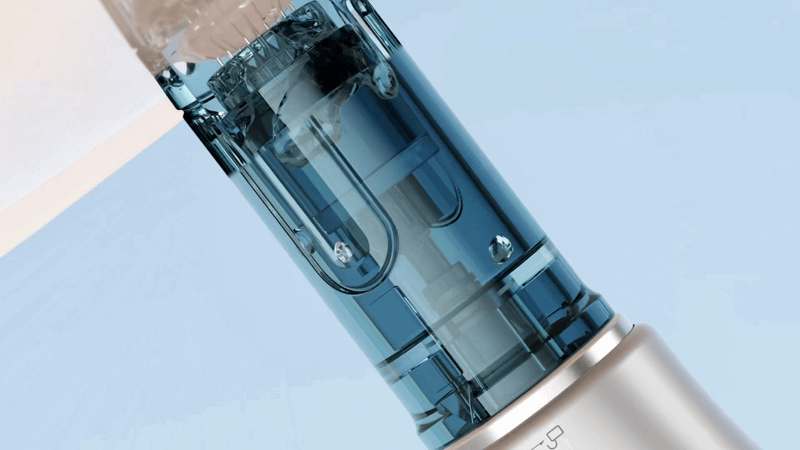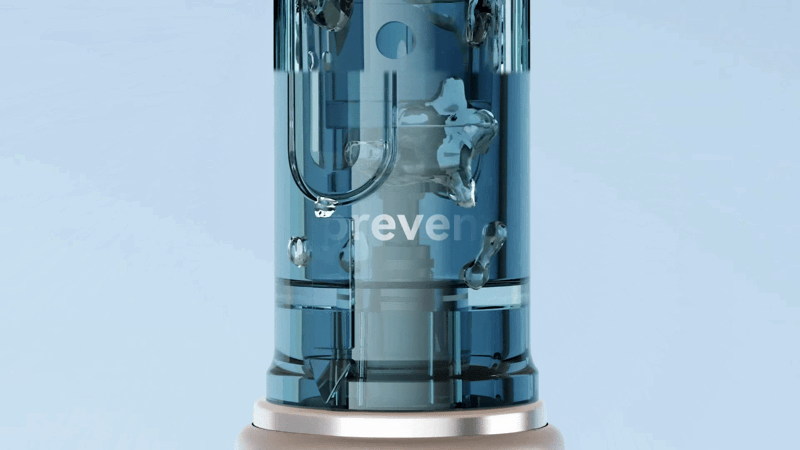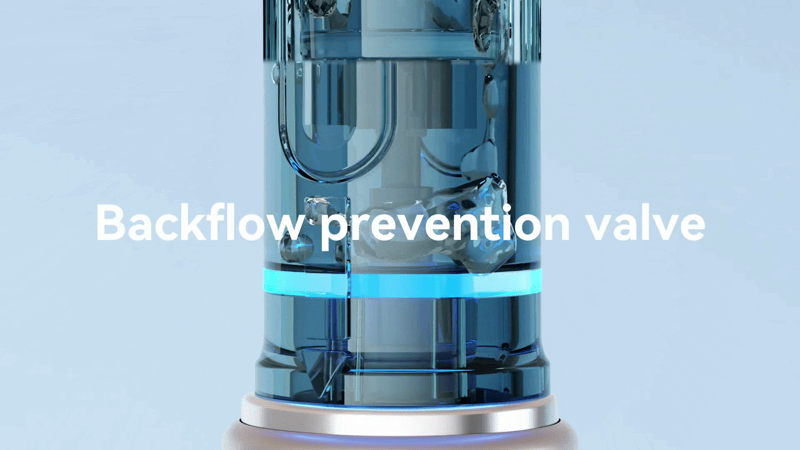
– Test Results: After approximately 10 minutes of inverted testing, inspection showed that the drive shaft connection area remained clean, with no red ink penetration. The liquid was completely contained outside the cartridge’s silicone sealing structure.


4. Evaluation Conclusion: Backflow Prevention Performance Verification
Even with a backflow prevention design in place, proper usage remains essential: always use disposable cartridges and ensure the device is thoroughly cleaned after each use to further minimize potential risks from an operational standpoint.


Dr. Pen M9 achieves stable and reliable backflow prevention through its refined structural design, ensuring both device safety and operational stability.
When combined with proper usage practices and good cleaning habits, it helps reduce potential risks, extend the device’s lifespan, and make the treatment process safer and more consistent.

